


PhET Lab experiments represent real interactive, and research-based simulations of physical phenomena from the PhET™ project at the University of Colorado. For teachers and students around the world, the PhET project provides interactive simulations that are based on extensive education research and support more effective science education. Going beyond traditional educational resources, PhET simulations offer an intuitive, game-like environment where students can learn through scientist-like exploration, where dynamic visual representations make the invisible visible, and where science ideas are connected to real-world phenomena. Unlike some educational software, these simulations are free, easily translated into multiple languages, and available either online or offline. PhET simulations are very flexible tools that can be used in many ways. PhET offers tips for integrating these simulations into your class.
All PhET simulations are available for free from the PhET website. They are written in Java and Flash, and can be run using a standard web browser as long as Flash and Java are installed. The PhET project is hard at work increasing the accessibility of their simulations, and a number of PhET simulations have accessibility features.
There are 53 simulations in the PhET Chemistry Collection. Here are some highlights:
| Online Activity Labs: | Topics: |
|---|---|
Acid-Base Solutions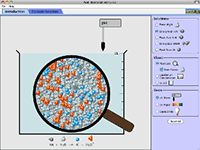 |
How do strong and weak acids differ? Use lab tools on your computer to find out! Dip the paper or the probe into solution to measure the pH, or put in the electrodes to measure the conductivity. Then see how concentration and strength affect pH. Can a weak acid solution have the same pH as a strong acid solution? Details and get access to Acid-Base Solutions Check out Acid-Base Solutions |
Build a Molecule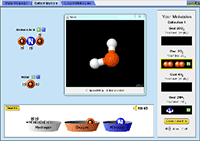 |
Starting from atoms, see how many molecules you can build. Collect your molecules and see them in 3D! Details and get access to Build a Molecule Check out Build a Molecule |
Molarity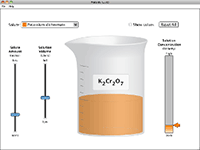 |
What determines the concentration of a solution? Learn about the relationships between moles, liters, and molarity by adjusting the amount of solute and solution volume. Change solutes to compare different chemical compounds in water.
Details and get access to Molarity Check out Molarity |
Molecule Shapes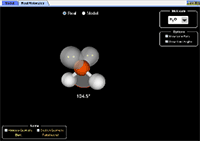 |
Explore molecule shapes by building molecules in 3D! How does molecule shape change with different numbers of bonds and electron pairs? Find out by adding single, double or triple bonds and lone pairs to the central atom. Then, compare the model to real molecules!
Details and get access to Molecule Shapes Check out Molecule Shapes |
Balancing Chemical Equations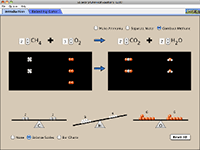 |
How do you know if a chemical equation is balanced? What can you change to balance an equation? Play a game to test your ideas! Details and get access to Balancing Chemical Equations Check out Balancing Chemical Equations |
| Website: | Topics: |
|---|---|
MERLOT |
| Website: | Topics: |
|---|---|
Open Text BC |
The BCcampus Open Education Virtual Lab and Science Resource Directory for Chemistry lists free chemistry resources designed to support remote chemistry education. |
| Website: | Topics: |
|---|---|
NSDL |
The National Science Digital Library - provides high quality online educational resources for teaching and learning, with current emphasis on the sciences, technology, engineering, and mathematics (STEM) disciplines–both formal and informal, institutional and individual, in local, state, national, and international educational settings. The NSDL collection contains structured descriptive information (metadata) about web-based educational resources held on other sites by their providers. These providers have contribute this metadata to NSDL for organized search and open access to educational resources via this website and its services. |
Are you using free Virtual Lab materials you found on the web in your teaching or your learning? Have you posted free Virtual Labs online that are open for others to use? We invite you to catalog these Virtual Labs you use or authored in MERLOT. Your colleagues and students around the world will thank you!
First: Become a member of MERLOT (It will take about 2 minutes and it’s FREE).
Second: Log in to MERLOT and click to Add a Material. (It will take about 4 minutes the first time and step-by-step instructions for contributing materials to MERLOT are available).